

Nutrition support for the critically ill is critical
How does malnutrition impact critically ill patients?
Several studies have observed a relationship between malnutrition and increased risk of mortality, infections, hospital length of stay.1-8 And unfortunately, many critically ill patients—those with cancer, Crohn’s disease, and short bowel syndrome just to name a few—often become malnourished. These conditions can affect their digestive system or interrupt their bowel function for a long time—or indefinitely. When this happens, clinical nutrition is needed to adequately nourish these patients.
More than 30% of patients in the intensive care unit have been reported to be malnourished.9
PN in critical care
Many critically ill patients cannot tolerate food by mouth or enteral feedings. In these cases, PN plays a crucial role in helping them meet their nutritional goals. In addition to amino acids, lipids, and dextrose, essential fatty acids (EFAs) are…well…essential to providing optimal nutrition.
The good news is that most lipid-based PN products provide EFAs. Lipid sources used in PN include soybean oil, medium-chain triglycerides (MCTs), olive oil, and fish oil. With SMOFlipid® (Lipid Injectable Emulsion, USP 20%), you get all four—each with a unique characteristic.
-

What makes SMOFlipid (Lipid Injectable Emulsion, USP 20%) unique?
SMOFlipid is the FIRST and ONLY four-oil lipid injectable emulsion (ILE) with demonstrated safety and tolerability10,11 in more than 7 million patients worldwide.* This unique combination of oils provides a balanced blend for critically ill patients of all ages—from adults to teenagers to preterm neonates.12
*Data on file.
What are the benefits of the blend?
-

Soybean oil 30% (omega-6)
Provides essential fatty acids.
-

Medium-chain triglycerides 30%
A source of rapidly available energy.13
-

Olive oil 25% (omega-9)
Supplies monounsaturated fatty acids.
-

Fish oil 15% (omega-3)
A source of EPA
and DHA.14
Studies showed triglyceride levels increased less with SMOFlipid compared to lipid emulsions with higher soybean oil content in adults.15,16
Triglyceride Change After 5 Days
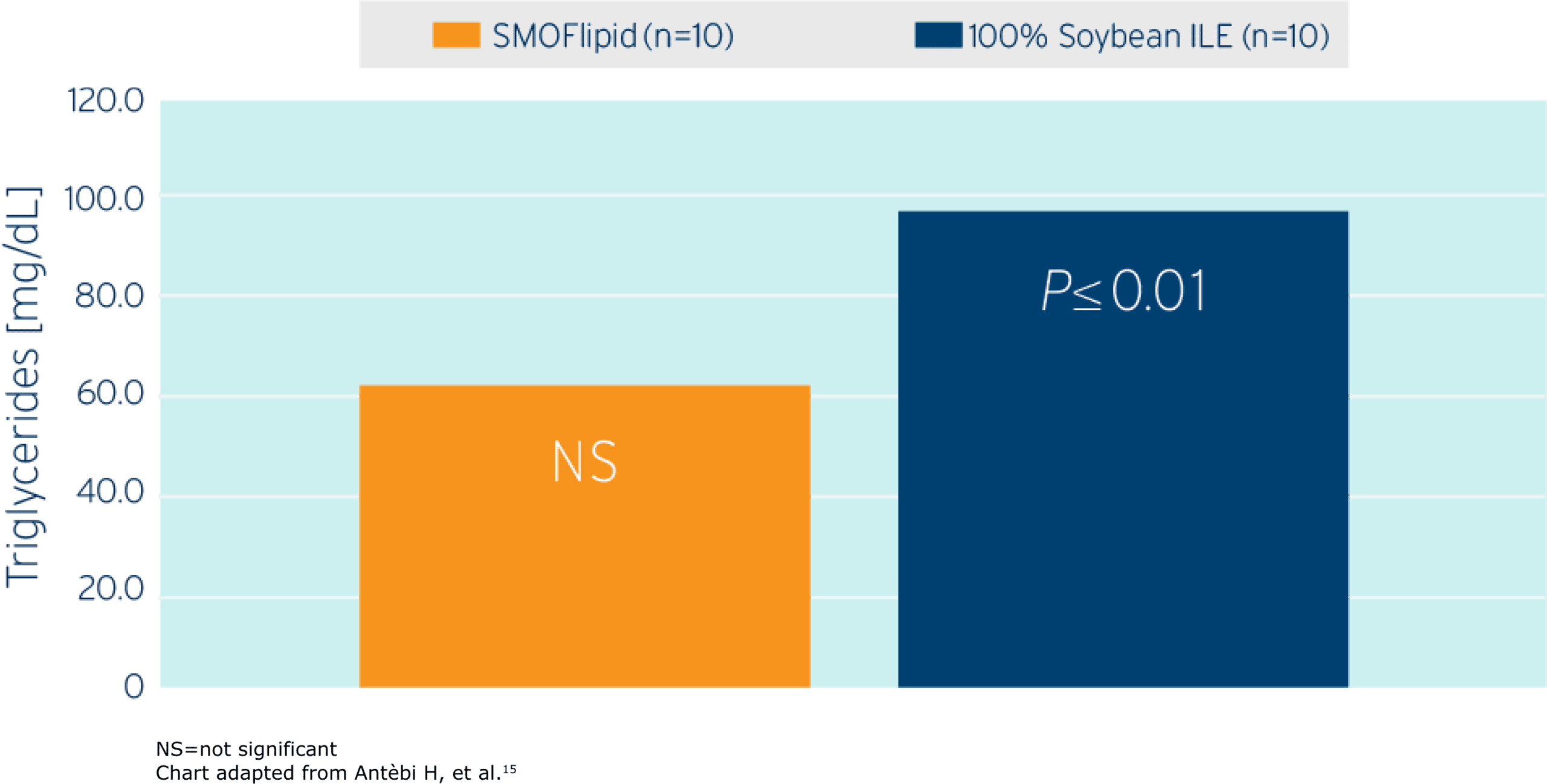
Study Takeaway
In this randomized controlled trial including postoperative surgical ICU adult patients (n=20), SMOFlipid demonstrated a lower triglyceride increase compared to those patients who received a lipid emulsion (1.5 g/kg/d ILE dose in both groups) with a higher soybean oil content.15
Monitor serum triglycerides before and during treatment with SMOFlipid. Company-sponsored studies showed that mean triglyceride levels from baseline values to week 4 were similar in both the SMOFlipid and comparator groups.
In pediatric patients, SMOFlipid has been shown to support growth and provides essential fatty acids.12,17,18
With SMOFlipid, Fresenius Kabi answered the call from PN and critical care medical societies for an alternative to soy-based ILEs.
As pioneers in clinical nutrition, Fresenius Kabi is committed to bringing innovations like SMOFlipid to market. Learn more about this unique 4-oil ILE and how it can help nourish your critically ill patients of any age at www.FreseniuKabiNutrition.com/products/smoflipid-adults/
SMOFLIPID (lipid injectable emulsion), for intravenous use
BRIEF SUMMARY OF PRESCRIBING INFORMATION
This brief summary does not include all the information needed to use SMOFlipid safely and effectively. Please see full prescribing information for intravenous use at www.FreseniusKabiNutrition.com/SMOFlipidPI.
INDICATIONS AND USAGE
SMOFlipid is indicated in adult and pediatric patients, including term and preterm neonates, as a source of calories and essential fatty acids for parenteral nutrition (PN) when oral or enteral nutrition is not possible, insufficient, or contraindicated.
DOSAGE AND ADMINISTRATION
The recommended daily dosage and initial and maximum infusion rates for pediatric and adult patients are provided in Table 1. Do not exceed the recommended maximum infusion rate in Table 1. The recommended duration of infusion for SMOFlipid will vary depending on the clinical situation. Adjust the administration flow rate by taking into account the dose being administered, the daily volume/intake, and the duration of the infusion.
SMOFlipid 1000 mL is supplied as a Pharmacy Bulk Package for admixing only and is not for direct infusion. Prior to administration, transfer to a separate PN container for individual patient use. Use a non-vented, non-DEHP 1.2 micron in-line filter during administration. Protect the admixed PN solution from light.
Table 1: Recommended Pediatric and Adult Dosage and Infusion Rate
| Age | Nutritional Requirements | Direct Infusion Rate | |
|---|---|---|---|
| Recommended Initial Dosage and Maximum Dosage | Initial | Maximum | |
| Birth to 2 years of age (including preterm and term neonates*) | Initial 0.5 to 1 g/kg/day not to exceed 3 g/kg/day** |
0.1 to 0.2 mL/kg/hour for the first 15 to 30 minutes; gradually increase to the required rate after 30 minutes | 0.75 mL/kg/hour |
| Pediatric patients 2 to <12 years of age | Initial 1 to 2 g/kg/day not to exceed 3 g/kg/day** |
0.2 to 0.4 mL/kg/hour for the first 15 to 30 minutes; gradually increase to the required rate after 30 minutes | 0.75 mL/kg/hour |
| Pediatric patients 12 to 17 years of age | Initial 1 g/kg/day not to exceed 2.5 g/kg/day** |
0.2 to 0.4 mL/kg/hour for the first 15 to 30 minutes; gradually increase to the required rate after 30 minutes | 0.75 mL/kg/hour |
| Adults | 1 to 2 g/kg/day not to exceed 2.5 g/kg/day** |
0.2 mL/kg/hour for the first 15 to 30 minutes; gradually increase to the required rate after 30 minutes | 0.5 mL/kg/hour |
*The neonatal period is defined as including term, post-term, and preterm newborn infants. The neonatal period for term and post-term infants is the day of birth plus 27 days. For preterm infants, the neonatal period is defined as the day of birth through the expected age of delivery plus 27 days (i.e., 44 weeks post-menstrual age).
** Daily dosage should not exceed a maximum of 60% of total energy requirements
CONTRAINDICATIONS
- Known hypersensitivity to fish, egg, soybean, peanut or to any of the active or inactive ingredients in SMOFlipid.
- Severe disorders of lipid metabolism characterized by hypertriglyceridemia (serum triglycerides >1,000 mg/dL).
WARNINGS AND PRECAUTIONS
-
Clinical Decompensation with Rapid Infusion of Intravenous Lipid Emulsion in Neonates and Infants.
In the postmarketing setting, serious adverse reactions including acute respiratory distress, metabolic acidosis, and death have been reported in neonates and infants after rapid infusion of intravenous lipid emulsions. Hypertriglyceridemia was commonly reported.
Strictly adhere to the recommended total daily dosage; the hourly infusion rate should not exceed 0.75 mL/kg/hour.
Preterm and small for gestational age infants have poor clearance of intravenous lipid emulsion and increased free fatty acid plasma levels following lipid emulsion infusion.
Carefully monitor the infant’s ability to eliminate the infused lipids from the circulation (e.g., measure serum triglycerides and/or plasma free fatty acid levels). If signs of poor clearance of lipids from the circulation occur, stop the infusion and initiate a medical evaluation.
-
Parenteral Nutrition-Associated Liver Disease and Other Hepatobiliary Disorders.
Risk of Parenteral Nutrition-Associated Liver Disease (PNALD): PNALD, or Intestinal failure-associated liver disease (IFALD), can present as cholestasis or hepatic stenosis and may progress to steatohepatitis with fibrosis and cirrhosis (possibly leading to chronic hepatic failure). The etiology of PNALD is multifactorial; however, intravenously administered phytosterols (plant sterols) contained in plant-derived lipid emulsions, including SMOFlipid, have been associated with development of PNALD.
In a randomized study of neonates and infants expected to be treated with PN for at least 28 days, parenteral nutrition-associated cholestasis (PNAC), a precursor to PNALD, developed less frequently in SMOFlipid-treated patients than in 100% soybean oil lipid emulsion-treated patients.
Monitor liver tests in patients treated with SMOFlipid and consider discontinuation or dosage reduction if abnormalities occur.
Other Hepatobiliary Disorders
Hepatobiliary disorders including cholecystitis and cholelithiasis have developed in some parenteral nutrition-treated patients without preexisting liver disease. Monitor liver tests when administering SMOFlipid. Patients developing signs of hepatobiliary disorders should be assessed early to determine whether these conditions are related to SMOFlipid use.
- Hypersensitivity Reactions: SMOFlipid contains soybean oil, fish oil, and egg phospholipids, which may cause hypersensitivity reactions. Cross reactions have been observed between soybean and peanut. SMOFlipid is contraindicated in patients with known hypersensitivity to fish, egg, soybean, peanut, or any of the active or inactive ingredients in SMOFlipid. If a hypersensitivity reaction occurs, stop infusion of SMOFlipid immediately and initiate appropriate treatment and supportive measures.
- Infections: Lipid emulsions, such as SMOFlipid, can support microbial growth and are an independent risk factor for the development of catheter-related bloodstream infections. To decrease the risk of infectious complications, ensure aseptic techniques are used for catheter placement, catheter maintenance, and preparation and administration of SMOFlipid. Monitor for signs and symptoms of infection including fever and chills, as well as laboratory test results that might indicate infection (including leukocytosis and hyperglycemia). Perform frequent checks of the intravenous catheter insertion site for edema, redness, and discharge.
-
Fat Overload Syndrome: This is a rare condition that has been reported with intravenous lipid emulsions and is characterized by a sudden deterioration in the patient’s condition (e.g., fever, anemia, leukopenia, thrombocytopenia, coagulation disorders, hyperlipidemia, hepatomegaly, deteriorating liver function, and central nervous system manifestations such as coma). A reduced or limited ability to metabolize lipids, accompanied by prolonged plasma clearance (resulting in higher lipid levels), may result in this syndrome. Although fat overload syndrome has been most frequently observed when the recommended lipid dose or infusion rate was exceeded, cases have also been described when the lipid formulation was administered according to instructions.
If signs or symptoms of fat overload syndrome occur, stop SMOFlipid. The syndrome is usually reversible when the infusion of the lipid emulsion is stopped.
- Refeeding Syndrome: Administering PN to severely malnourished patients may result in refeeding syndrome, which is characterized by the intracellular shift of potassium, phosphorus, and magnesium as patients become anabolic. Thiamine deficiency and fluid retention may also develop. To prevent these complications, closely monitor severely malnourished patients and slowly increase their nutrient intake.
-
Hypertriglyceridemia: The use of SMOFlipid is contraindicated in patients with hypertriglyceridemia with serum triglyceride concentrations >1,000 mg/dL.
Patients with conditions such as inherited lipid disorders, obesity, diabetes mellitus, or metabolic syndromes have a higher risk of developing hypertriglyceridemia with the use of SMOFlipid. In addition, patients with hypertriglyceridemia may have worsening of their hypertriglyceridemia with administration of SMOFlipid. Excessive dextrose administration may further increase such risk.
Evaluate patients’ capacity to metabolize and eliminate the infused lipid emulsion by measuring serum triglycerides before the start of infusion (baseline value) and regularly throughout treatment. If triglyceride levels are above 400 mg/dL in adults, stop the SMOFlipid infusion and monitor serum triglyceride levels to avoid clinical consequences of hypertriglyceridemia such as pancreatitis. In pediatric patients with hypertriglyceridemia, lower triglyceride levels (i.e., below 400 mg/dL) may be associated with adverse reactions. Monitor serum triglyceride levels to avoid potential complications with hypertriglyceridemia such as pancreatitis, lipid pneumonitis, and neurologic changes, including kernicterus.
To minimize the risk of new or worsening of hypertriglyceridemia, assess high-risk patients for their overall energy intake including other sources of lipids and dextrose, as well as concomitant drugs that may affect lipid and dextrose metabolism.
- Aluminum Toxicity: SMOFlipid contains no more than 25 mcg/L of aluminum. Prolonged PN administration in patients with renal impairment may result in aluminum reaching toxic levels. Preterm infants are at greater risk because their kidneys are immature, and they require large amounts of calcium and phosphate solutions, which contain aluminum. Patients with impaired kidney function, including preterm infants, who receive parenteral levels of aluminum at greater than 4 to 5 mcg/kg/day can accumulate aluminum at levels associated with central nervous system and bone toxicity. Tissue loading may occur at even lower rates of administration.
- Essential Fatty Acid Deficiency: Treatment-emergent cases of moderate or severe essential fatty acid deficiency (EFAD) (defined as the triene [Mead acid] to tetraene [arachidonic acid] ratio >0.2 and >0.4, respectively) were not observed in pediatric clinical trials of SMOFlipid up to 28 days. However, cases of EFAD have been reported in adults and pediatric patients in the postmarketing period with the use of SMOFlipid. The median time to onset was greater than 28 days among cases that reported time to onset. Monitor patients for laboratory evidence (e.g., abnormal fatty acid levels) and clinical symptoms of EFAD (e.g., skin manifestations and poor growth) because these signs may emerge before laboratory evidence of EFAD is confirmed. Laboratory testing using the triene to tetraene ratio may not be adequate to diagnose EFAD, and assessment of individual fatty acid levels may be needed. Ensure patients are receiving recommended dosages of SMOFlipid to prevent EFAD.
-
Monitoring/Laboratory Tests: Throughout treatment monitor serum triglycerides, fluid and electrolyte status, blood glucose, liver and kidney function, coagulation parameters, and complete blood count including platelets.
The lipids contained in SMOFlipid may interfere with some laboratory blood tests (e.g., hemoglobin, lactate dehydrogenase [LDH], bilirubin, and oxygen saturation) if blood is sampled before lipids have cleared from the bloodstream. Conduct these blood tests at least 6 hours after stopping the infusion. SMOFlipid contains vitamin K that may counteract anticoagulant activity.
ADVERSE REACTIONS
Most common adverse drug reactions >1% of adult patients who received SMOFlipid from clinical trials were nausea, vomiting, hyperglycemia, flatulence, pyrexia, abdominal pain, increased blood triglycerides, hypertension, sepsis, dyspepsia, urinary tract infection, anemia, and device-related infection.
Less common adverse reactions in ≤1% of adult patients who received SMOFlipid were dyspnea, leukocytosis, diarrhea, pneumonia, cholestasis, dysgeusia, increased blood alkaline phosphatase, increased gamma-glutamyltransferase, increased C-reactive protein, tachycardia, liver function test abnormalities, headache, pruritis, dizziness, rash, and thrombophlebitis.
The most common adverse drug reactions in >1% of pediatric patients who received SMOFlipid were anemia, vomiting, gamma-glutamyltransferase increased, nosocomial infection, cholestasis, pyrexia, C-reactive protein increased, hyperbilirubinemia, abdominal pain, bilirubin conjugated increased, diarrhea, tachycardia, thrombocytopenia, hyperglycemia, and sepsis.
Less common adverse reactions in ≤1% of pediatric patients who received SMOFlipid were decreased hematocrit, metabolic acidosis, increased blood triglycerides, infection, increased blood alkaline phosphatase, increased alanine aminotransferase, fluid overload, hypertension, hypertriglyceridemia, and rash.
The following adverse reactions have been identified during post-approval use of SMOFlipid in countries where it is registered. Cardiac disorders: palpitations; General disorders and administration site conditions: chills, chest pain, malaise; Hepatobiliary disorders: cholestasis; Infections and infestations: infection; Metabolism and nutrition disorders: fatty acid deficiency; Respiratory, thoracic and mediastinal disorders: dyspnea; Skin and subcutaneous tissue disorders: hyperhidrosis; Vascular disorders: phlebitis.
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176, option 5, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Soybean and olive oils in SMOFlipid contain vitamin K1 which may counteract the anticoagulant activity of vitamin K antagonists such as warfarin. In patients who receive concomitant SMOFlipid and warfarin, increase monitoring of laboratory parameters for anticoagulant activity.
USE IN SPECIFIC POPULATIONS
- Pregnancy and Lactation: Administration of the recommended dose of SMOFlipid is not expected to cause major birth defects, miscarriage, or other adverse maternal or fetal outcomes. No animal reproduction studies have been conducted with SMOFlipid. Administration of the recommended dose of SMOFlipid is not expected to cause harm to a breastfed infant. There are no data on the presence of SMOFlipid in human or animal milk or its effects on milk production.
- Pediatric Use: The safety and effectiveness of SMOFlipid have been established as a source of calories and essential fatty acids for parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated in pediatric patients, including term and preterm neonates. Use of SMOFlipid in neonates is supported by evidence from short-term (i.e., 1- to 4-week) studies, and one study following neonates beyond 4 weeks. Use of SMOFlipid in older pediatric patients is supported by evidence from a short-term (i.e., <28 days) study in pediatric patients 28 days to 12 years of age and additional evidence from studies in adults. The most common adverse reactions in SMOFlipid-treated pediatric patients were anemia, vomiting, gamma-glutamyltransferase increased, and nosocomial infection. PNALD, also referred to as IFALD, has been reported in pediatric patients who received SMOFlipid for more than 2 weeks. PNAC (a precursor to PNALD) was reported less frequently in SMOFlipid-treated patients compared to soybean oil lipid emulsion-treated patients in Pediatric Study 1. Although clinically significant cases of EFAD were not observed during short-term use in pediatric clinical studies, cases of EFAD have been reported with the use of SMOFlipid in the postmarketing setting. Monitor pediatric patients for laboratory evidence of EFAD because they may be particularly vulnerable to neurologic complications if adequate amounts of essential fatty acids are not provided. In the post marketing setting, clinical decompensation with rapid infusion of intravenous lipid emulsion in neonates and infants, sometimes fatal has been reported. Because of immature renal function, preterm infants receiving prolonged treatment with SMOFlipid may be at risk for aluminum toxicity.
- Geriatric Use: Energy expenditure and requirements may be lower for older adults than younger patients. Of the 354 adult patients in clinical studies of SMOFlipid, 35% were >65 years of age and 10% were >75 years of age. No overall differences in the safety and efficacy of SMOFlipid were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity in some older patients cannot be ruled out.
OVERDOSAGE
In the event of an overdose, serious adverse reactions may result. Stop the SMOFlipid infusion until triglyceride levels have normalized and symptoms have abated. The effects are usually reversible by stopping the lipid infusion. If medically appropriate, further intervention may be indicated. Lipids are not dialyzable from plasma.
3673-SMF-08-06/23